The topic of ring structure and isomerism in monosaccharides is one of the most troublesome subjects in food chemistry because it contains a great deal of detail, includes many terms, and, in particular, because the different isomers are very often confused with one another.
Given the importance of the subject, in this article I will try to explain both ring structures and the isomerism of monosaccharides in a detailed and understandable way. In the first part of the article, the ring structure of monosaccharides is explained. In the second part, which appears further below, I will try to explain isomerism in monosaccharides and the classification of this isomerism.
1. The Ring Structure of Monosaccharides
In the Fischer projection, the structure of monosaccharides appears to be in the form of a straight chain (see Carbohydrates; Monosaccharides, Disaccharides and Polysaccharides). However, although this theoretical straight-chain structure is correct, monosaccharides containing 5 or more carbon (C) atoms in their chain are found in ring form under natural conditions.
As is known, monosaccharides -whether aldoses or ketoses- contain one carbonyl group (C=O) and more than one hydroxyl group (-OH) in their structure. As may be recalled from organic chemistry, the carbonyl group is electrophilic.
Under these circumstances, one of the hydroxyl groups present in the structure of monosaccharides (generally the -OH groups attached to the C5 or C4 carbon atoms) makes a nucleophilic attack on the carbonyl group in the same chain (the lone pair of electrons on the oxygen atom of the -OH group attacks the carbon atom of the carbonyl group).
As a result, a covalent bond (C–O) forms between the oxygen attached to the hydroxyl group and the carbon atom of the carbonyl group, and the straight-chain structure is converted into a ring structure.
To describe it in the language of chemistry, the carbonyl group and the hydroxyl group undergo an intramolecular reaction, forming a hemiacetal or a hemiketal.
In the Fischer projection, the D and L forms of monosaccharides can be shown easily in the straight-chain structure. However, Haworth formulas are used in order to show the ring structure and the newly formed stereoisomeric forms in an understandable way. Haworth formulas have been used for more than 100 years to represent the three-dimensional ring structure clearly on paper. In the Haworth formula, the plane is considered horizontal. The bonds drawn with thicker lines than the others indicate that that part of the molecule is closer to the viewer.
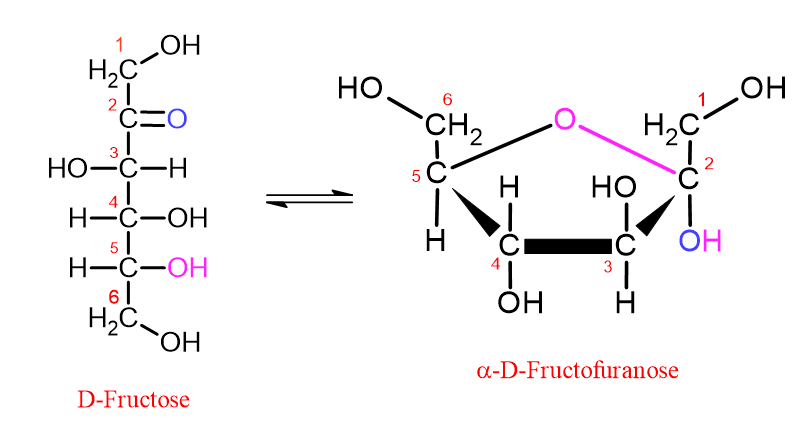
In general, reference sources explain the ring structures of monosaccharides with statements such as “monosaccharides with 5 or more carbon atoms show a ring structure in aqueous media or in solutions.” However, this explanation is quite prone to misunderstanding and is misleading in the meaning it suggests.
Because from this statement one may infer that monosaccharides exhibit a ring structure only in aqueous environments, whereas in crystalline and dry form they remain in the open straight-chain form. In fact, monosaccharides with 5 or more carbon atoms exist in ring form under almost all conditions and in almost all environments. To take glucose as an example, glucose is produced in the plant in the open-chain form, but in the cellular environment it assumes the ring structure immediately, within milliseconds after its formation.
On the other hand, the open straight-chain form and the ring form of monosaccharides are in a constant state of interconversion. For example, in an aqueous solution of glucose, the glucose molecule continuously shifts back and forth between the open straight-chain form and the ring form. However, at any given moment, more than 99% of the glucose in solution is in the ring form, while less than 0.1% of the remainder is in the open straight-chain form. In the dry crystalline state of glucose, nearly all of the molecules are in the ring form, and a transition to the open straight-chain form, as seen in aqueous media, does not occur. In larger structures such as sucrose and starch, glucose is again present in ring form.
Several questions may arise here. Let us try to answer them briefly:
Question 1: Why can monosaccharides with 5 or more carbon atoms form ring structures, whereas monosaccharides with fewer carbon atoms cannot?
Answer: This is entirely related to stability and suitability. Monosaccharides with 3 carbon atoms are not long enough to form a ring structure. In monosaccharides with 4 carbon atoms, the ring structure that would form would not be stable because of high intramolecular strain.
Question 2: Why does the hydroxyl group not attack the carbonyl group of a neighboring monosaccharide, but instead attack its own carbonyl group?
Answer: The most important reason is the lack of effective concentration. Within the molecule, the carbonyl and hydroxyl groups are always close to each other and are in the correct geometric position. For a group on another molecule to be nearby, at the correct angle, and at the correct energy is something that must be awaited; this is quite a significant disadvantage. In addition, if such a bond were to form, a very large and irregular molecule would result, which is not a favorable pathway.
In monosaccharides with more than 5 carbon atoms, there are two hydroxyl groups (-OH) at a suitable distance to bond to the carbon atom of the carbonyl group (C=O) during ring formation: the hydroxyl groups attached to C4 and C5.
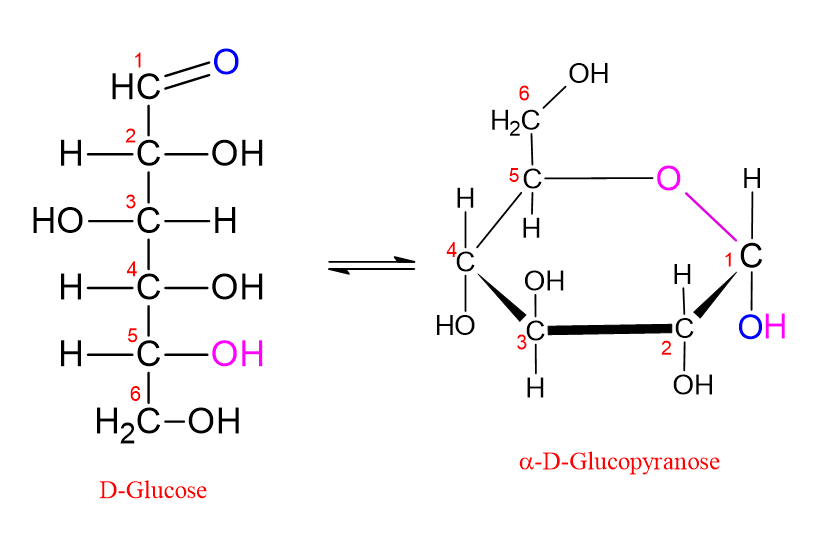
Let us explain this through glucose, which contains 6 carbon atoms in its structure: in glucose, the carbonyl group (C=O) is located on the first carbon (C1). If the hydroxyl group attached to the C5 carbon in the chain attacks the carbonyl carbon, a six-membered ring is formed. This is called a “pyranose ring.” This name was given because it resembles the pyran molecule, which is a six-membered oxygen-containing ring structure.
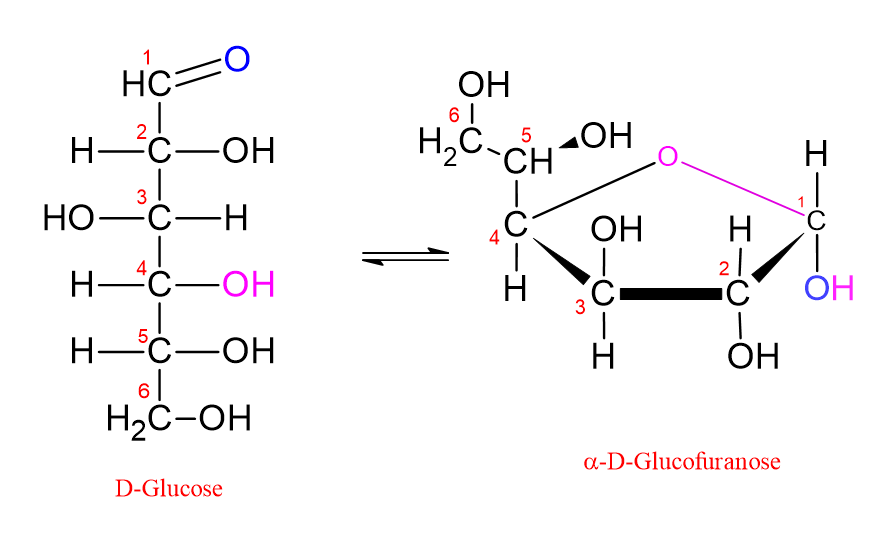
By contrast, the hydroxyl group on the C4 carbon of glucose is also at a suitable distance to react with the carbon at C1. If the hydroxyl group attached to the C4 carbon attacks the carbonyl carbon, then a five-membered ring is formed. This is called a “furanose ring.” This name was given because it resembles the furan molecule, which is a five-membered oxygen-containing ring structure.
As a result, two different ring types can be seen in monosaccharides:
1. Pyranose; a six-membered ring structure in which one of the members is oxygen
2. Furanose; a five-membered ring structure in which one of the members is oxygen
The pyranose and furanose ring structures of some monosaccharides are shown below;
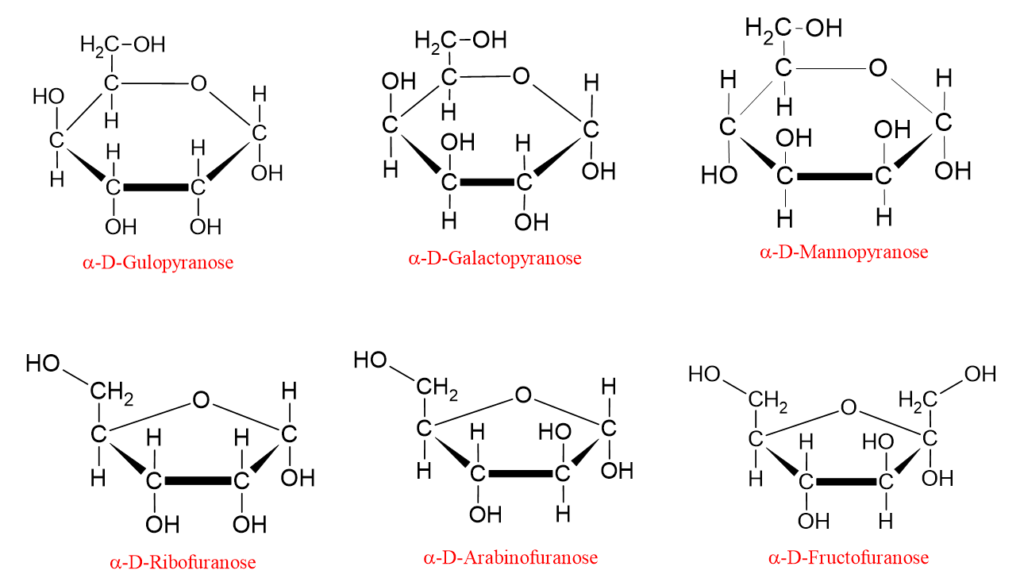
At this point, the following question may come to mind: why are only 5- and 6-membered ring structures seen in monosaccharides?
Answer: During ring formation in monosaccharides, rings of different sizes can theoretically form. However, the rings most commonly encountered in nature are five- and six-membered rings. The main reason for this is that rings of these sizes are chemically quite stable.
As may be recalled from organic chemistry, ring stability is largely related to bond angles and intramolecular strain. When carbon atoms have sp³ hybridization, the ideal bond angle is approximately 109.5°. In very small rings, this angle is seriously distorted, and high strain arises within the molecule.
For example, in three- or four-membered rings, the bond angles deviate considerably from the ideal value. For this reason, such rings have high energy and are unstable. By contrast, in five- and six-membered rings, the bond angles are closer to the ideal, and intramolecular strain is much lower. Therefore, these rings are regarded as more stable.
In rings larger than six members, bond-angle strain is generally not a major problem. However, when these rings form, it becomes more difficult for the chain to close in the correct position. In other words, the probability that the ends of the molecule will come together at the correct position and angle decreases. This makes ring formation kinetically less favorable. In addition, because large rings are generally more flexible, additional strains may also arise in certain conformations.
To state it briefly:
• Rings with fewer than 5 members are not stable because of angle strain.
• Five- and six-membered rings are the most stable ring structures.
• Rings with more than 6 members have a low probability of closure and are prone to additional strains.
The table below presents the general tendency regarding which ring structure is predominant in which monosaccharides;
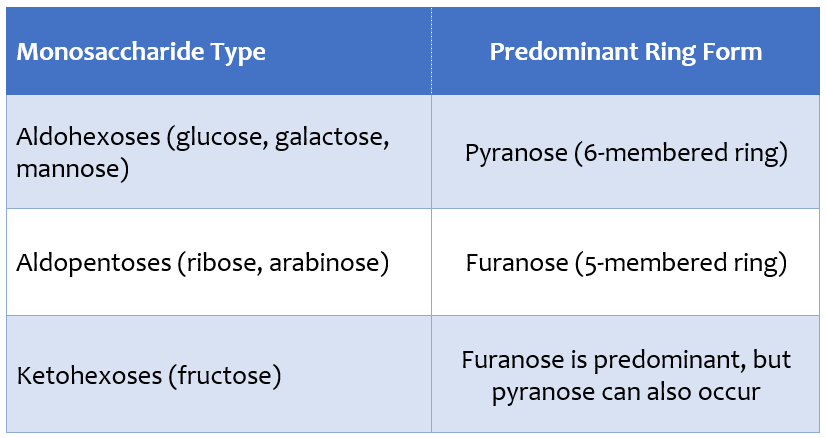
This table is not an absolute rule, but it does show a strong tendency. In solution, monosaccharides exist in a state of dynamic equilibrium between different ring forms. Therefore, in addition to the predominant ring form, other ring types may also be present in small proportions. However, in most monosaccharides, this equilibrium clearly favors a single ring form.
The ring structure also brings with it a new chiral center. Continuing with glucose as an example, when glucose forms a pyranose structure, the C1 carbon that contains the carbonyl group also becomes a new chiral center (also referred to as an asymmetric carbon or the anomeric carbon).
In the straight-chain structure of glucose, the chirality of the carbons is as follows:
• C1: not chiral
• C2: chiral center
• C3: chiral center
• C4: chiral center
• C5: chiral center
• C6: not chiral
When determining the configuration of the molecule in the straight-chain structure, the chiral carbon with the highest carbon number is taken as the basis. In this case, if the hydroxyl group (-OH) on the C5 carbon of glucose is on the right, it is expressed as the D configuration; if it is on the left, it is expressed as the L configuration.
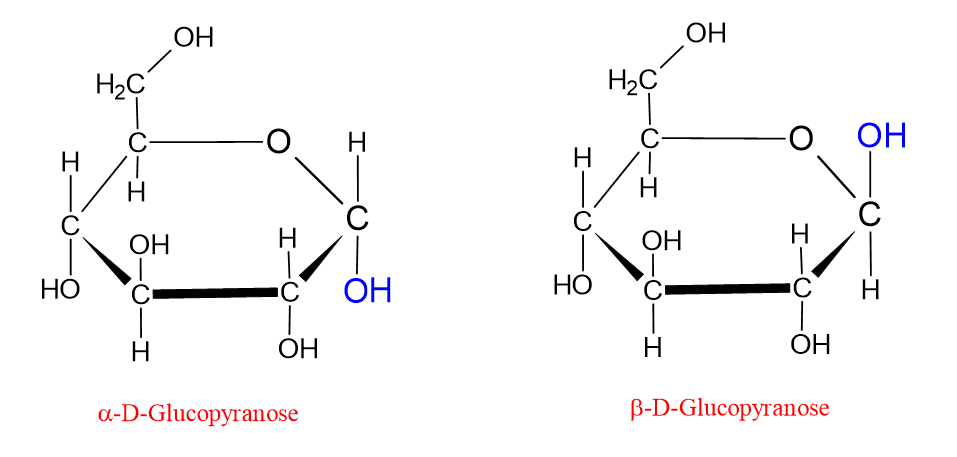
However, with the transition to the ring structure, the C1 carbon also becomes chiral (this carbon is now called the anomeric carbon). Therefore, two additional configuration possibilities arise in the ring structure. Depending on the position of the hydroxyl group (-OH) attached to this newly formed chiral carbon atom, two new stereoisomeric forms occur in the ring structure; α (alpha) and β (beta).
In the Fischer projection, the hydroxyl groups shown on the right side of the molecule are shown on the lower side of the molecule in the Haworth formula. Those shown on the left side in the Fischer projection are shown on the upper side in the Haworth formula. In the Haworth formula, if the hydroxyl group attached to the anomeric carbon (C1) is shown below, the molecule is called the α form. If the hydroxyl group attached to the anomeric carbon (C1) is shown above, the molecule is called the β form. This is shown in the figure below;
Haworth projections are highly useful for showing the ring structure of monosaccharides clearly. However, this representation does not fully reflect the true three-dimensional geometry of the molecule. In the Haworth formula, the ring is drawn like a flat pentagon or hexagon. In reality, however, carbon atoms have sp³ hybridization and bond angles of approximately 109.5°. For this reason, molecules cannot form a perfectly flat ring structure. In order to bring the bond angles closer to the ideal and reduce intramolecular strain, the molecule adopts certain conformations in space.
Five-membered furanose rings generally adopt envelope and twist conformations, whereas six-membered pyranose rings are found in nature mostly in chair and boat conformations. The main reason for the emergence of these conformations is to bring the bond angles closer to the ideal tetrahedral angle and to reduce intramolecular strain. The different conformations formed by monosaccharides are given below;
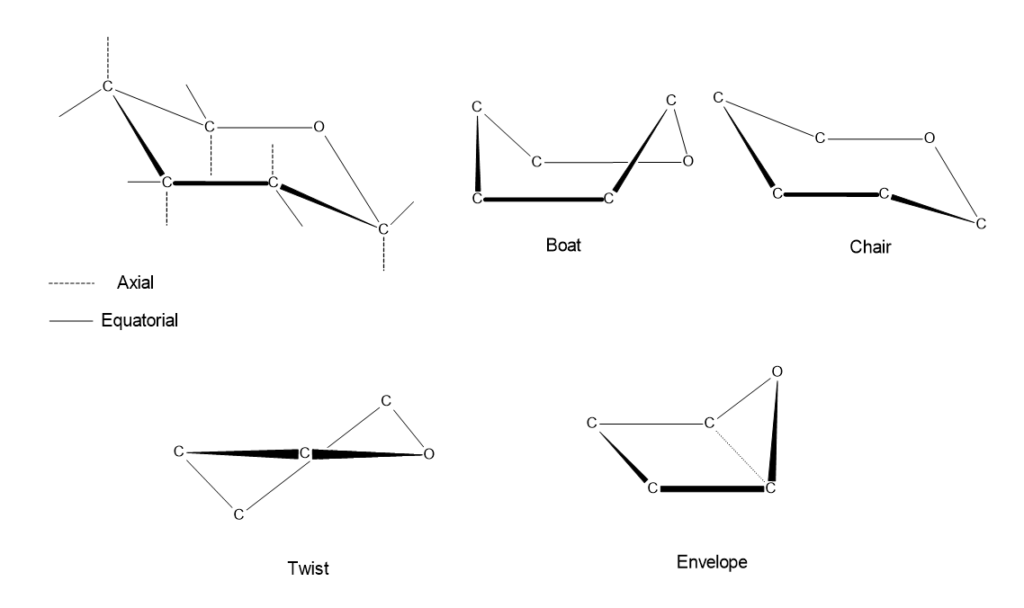
In the chair conformation, the groups attached to each carbon atom in the ring can occupy two different positions in space; axial and equatorial. Axial bonds are oriented approximately perpendicular to the ring, either upward or downward. Equatorial bonds extend outward from the ring in a direction approximately parallel to the plane of the ring.
In general, the equatorial position is more stable. The reason is that large groups in the axial position create steric repulsion with other axial groups on the ring (1,3-diaxial interactions). For this reason, small atoms such as hydrogen may occupy the axial position, but larger groups prefer the equatorial position.
In summary, it may be stated as follows: in the chair conformation of six-membered monosaccharide rings, the preference of large groups for the equatorial position reduces intramolecular steric interactions and increases the stability of the structure.
In the chair conformation of β-D-glucopyranose, nearly all hydroxyl groups are in the equatorial position, while only the hydrogen atoms occupy axial positions. This greatly reduces intramolecular steric strain and makes the structure highly stable. Therefore, β-D-glucopyranose is the most stable form of glucose and is present in the highest proportion in solution.
At this point, we may say that we have completed the ring structure of monosaccharides. What remains is the explanation of optical rotation and mutarotation in monosaccharides.
1.1. Optical Rotation and Mutarotation in Monosaccharides
Because monosaccharides contain more than one chiral carbon atom, they are optically active. Optically active molecules have the property of rotating plane-polarized light in a certain direction. The phenomenon of changing the direction of polarized light is called optical rotation.
Two different stereoisomers of an optically active molecule may often exhibit similar physical and chemical properties. However, the direction and angle by which these two stereoisomers rotate polarized light are unique to them. Therefore, optical activity is frequently used to distinguish between the stereoisomers of a molecule.
If a molecule rotates polarized light clockwise, it is called positive (+) or dextrorotatory; if it rotates it counterclockwise, it is called negative (−) or levorotatory.
An important point here is that these signs cannot be reliably predicted by looking at the structure of the molecule. The direction in which a molecule rotates polarized light is generally determined experimentally by measurements made with an instrument called a polarimeter. Therefore, the (+) or (−) sign placed before a molecule’s name does not indicate a theoretical prediction, but rather an experimentally measured property.
Another important point regarding optical rotation is that it should not be confused with the D and L designation. The D and L system expresses the configuration of the molecule and is determined according to the position of a specific chiral carbon in the Fischer projection. By contrast, the (+) and (−) signs show the direction in which the molecule rotates polarized light. Therefore, there is no direct relationship between D or L configuration and the direction of optical rotation. For example, glucose is generally expressed as D-(+)-glucose, whereas fructose may be shown as D-(−)-fructose. As can be seen, although both molecules are in the D configuration, their directions of optical rotation are different.
Finally, when naming a molecule, writing the (+) or (−) sign is often not mandatory. In the identification of monosaccharides, indicating the D or L configuration is generally considered sufficient. However, in cases where the direction of optical rotation must be emphasized specifically, a (+) or (−) sign may be added before the name of the molecule. These signs express experimental information used to indicate the stereochemical behavior and optical properties of the molecule.
Monosaccharides such as glucose do not exist in only a single ring form in aqueous solution. The molecules continuously interconvert between the α and β anomers through repeated ring opening and closing. As a result of this process, the optical rotation value of the solution changes over time and eventually reaches a constant value. The phenomenon in which optical rotation changes over time and reaches an equilibrium value is called “mutarotation.”
The optical rotation of a freshly prepared α-D-glucose solution is approximately +112°. However, over time, the optical rotation of the solution decreases to +52.7° and becomes fixed at that point. This occurs for the following reason: the α-D-glucose in solution continuously undergoes ring opening, and when the ring closes again, the new structure of the molecule may be either α or β. β-D-glucose has an optical rotation of approximately +18.7°. Therefore, as the proportion of the β form of the molecule increases in solution, the degree of optical rotation of the solution decreases. When equilibrium is established between the two forms, the optical rotation of the solution becomes fixed at +52.7°. From this equilibrium optical rotation value, it is calculated that the solution contains approximately 64% β-D-glucopyranose and approximately 36% α-D-glucopyranose.
As a result, while optical rotation reveals the chiral structure and stereochemical properties of monosaccharides, mutarotation shows the dynamic equilibrium behavior of these molecules in solution. Together, these two properties play an important role in understanding the chemical and biological properties of monosaccharides.
2. Isomerism in Monosaccharides
The information acquired so far makes a general collective explanation of isomerism in monosaccharides necessary. This is because we have mentioned many different types of isomerism, and they are quite prone to being confused with one another.
Because monosaccharides contain more than one chiral carbon atom in their structure, they can form many isomers. Molecules that have the same molecular formula but differ in structure or in their spatial arrangement are called isomers. Isomers are generally divided into two main groups: structural (constitutional) isomers and stereoisomers.
2.1. Structural (Constitutional) Isomers
In structural isomers, the bonding arrangement between the atoms of the molecules is different. In other words, the way the atoms are connected to each other has changed. In monosaccharides, this situation generally arises from the different position of the carbonyl group.
For example, glucose and fructose have the same molecular formula (C₆H₁₂O₆). However, glucose is an aldose, whereas fructose is a ketose. Therefore, the position of the carbonyl group is different, and these two sugars are structural isomers.
2.2. Stereoisomers
In stereoisomers, the bonding arrangement of the molecules is the same, but the spatial arrangement of the atoms is different. Stereoisomers are divided into two main groups: enantiomers and diastereomers.
2.2.1. Enantiomers
Enantiomers are stereoisomers that are mirror images of one another. These molecules have the same physical properties in an achiral environment, that is, in the absence of a chiral reagent or chiral interaction in the environment, but they rotate plane-polarized light in opposite directions. In carbohydrates, enantiomerism is generally expressed by the D and L designation. For example, D-glucose and L-glucose are enantiomers of each other.
One point must be emphasized here in particular. The fact that only the highest-numbered chiral carbon atom in the molecule is considered when determining the D and L designation does not mean that the difference between the two molecules lies only in that carbon atom. The D and L system is merely a classification method. In this system, whether the molecule belongs to the D or L series is determined according to the position of the carbon atom used as the reference.
In reality, however, in two molecules that are enantiomers, the configuration of all chiral centers is the opposite of one another. For example, when D-glucose and L-glucose are compared, the difference is not only at the C5 carbon; the stereochemical arrangement at all of the C2, C3, C4, and C5 carbons is the mirror image of the other. However, in the D/L classification, only the highest-numbered chiral carbon is taken as the reference. Therefore, the D and L designation does not express all the stereochemical differences between the two molecules, but rather a practical naming system indicating only which series they belong to.
2.2.2. Diastereomers
Diastereomers are stereoisomers that are not mirror images of one another. The physical and chemical properties of these molecules are generally different.
2.2.2.1. Epimers
A special subgroup of diastereomers is called epimers. Epimers are diastereomers whose configuration differs at only a single chiral carbon atom. For example, D-glucose and D-mannose are C2 epimers of each other. D-glucose and D-galactose are C4 epimers of each other.
2.2.2.2. Anomers
When monosaccharides form a ring, the carbonyl carbon becomes a new chiral center. This carbon is called the anomeric carbon. The isomers formed depending on the configuration of the anomeric carbon are called anomers. For example, α-D-glucose and β-D-glucose are anomers of each other.
I usually explain isomerism in monosaccharides to my students as follows:
Think of a person’s two hands.
• Under normal conditions, the two hands have the same structure; both hands have fingers, and the points where the fingers are attached are the same, and the two hands are mirror images of one another. In this case, the two hands are enantiomers of each other. D-glucose and L-glucose are like this.
• Now imagine that in one of the two hands, the thumb does not emerge from its normal place, but instead comes out from the palm. In this case, the structure has now changed. The same palm, the same fingers, the same components; but in one hand, unlike the other, one of the fingers comes out from the palm. We can no longer speak of two hands that are mirror images of each other. In this case, these two hands are structural isomers of each other. Glucose and fructose are like this.
• Now imagine that in one of the two hands, the index finger is bent forward, while in the other hand, the index finger is bent backward. The same palm, the same fingers, and attached to the palm at the same points; however, the positions of the index fingers are different. In this case as well, these two hands are no longer mirror images of one another. These two hands are epimers of each other. Glucose and galactose are like this.
• Now imagine that in one of the two hands, the thumbnail is very long and bent forward, while in the other hand, the thumbnail is also very long but bent backward. In this case, everything is the same, but the directions of the thumbnails are different. These two hands are anomers of each other. α-D-glucose and β-D-glucose are like this.
In the table below, the classification of isomerism in monosaccharides and some important properties is presented;
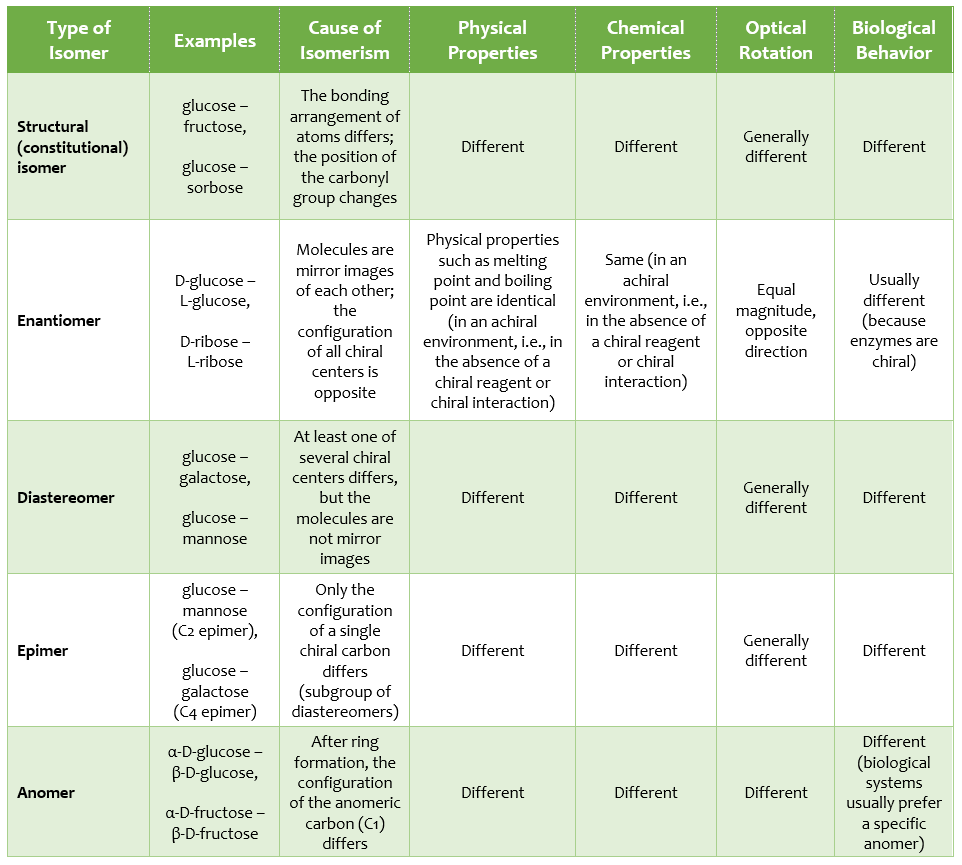
At this point, the following thought may arise: “Since glucose and galactose have the same molecular formula, they are structural isomers.” However, this thought is not correct. Because for a molecule to be considered a structural isomer, it is not enough for it merely to have the same molecular formula; the bonding arrangement of the atoms must also be different.
If the bonding arrangement of the atoms has changed between isomers, structural (constitutional) isomerism is involved; if the bonding arrangement remains the same and only the spatial arrangement of the atoms changes, stereoisomerism is involved. For example, although glucose and fructose have the same molecular formula (C₆H₁₂O₆), they are structural isomers because the position of the carbonyl group is different. By contrast, in glucose and galactose, the bonding arrangement of the atoms is the same; the difference arises only from the spatial arrangement of the hydroxyl groups at certain chiral carbons. Therefore, glucose and galactose are not structural isomers, but stereoisomers.
In short, “having the same molecular formula” alone does not mean that structural isomerism is present. What is decisive is how the atoms are bonded to one another.
Another important rule that determines the number of isomers in monosaccharides is the number of chiral carbon atoms the molecule possesses. If a molecule contains n chiral carbon atoms, the maximum number of stereoisomers that can theoretically form is 2ⁿ. The reason for this is that each chiral carbon atom can have two different configurations. For example, if a molecule has two chiral carbons, theoretically 2² = 4 different stereoisomers can form; if it has three chiral carbons, 2³ = 8 different stereoisomers can form. This rule also applies to monosaccharides.
For example, since aldohexoses have four chiral carbon atoms (C2, C3, C4, and C5) in their open-chain form, theoretically 2⁴ = 16 different stereoisomers can form. Half of these isomers belong to the D series and the other half belong to the L series. Glucose, galactose, and mannose are only a few examples representing these isomers. Therefore, the monosaccharides we encounter in nature represent only a small portion of the stereoisomers that are theoretically possible.