Carbohydrates are organic molecules consisting of carbon (C), hydrogen (H) and oxygen (O). Plants produce carbohydrates by using carbon dioxide and water through photosynthesis.
While some of the carbohydrates produced are converted into protein, fat and other organic compounds, some are turned into polysaccharides for storage.
Carbohydrates are consumed by organisms that cannot photosynthesize, primarily to obtain energy. Although it is a natural component in foods, some carbohydrates can be added to foods as gelling, stabilizers and thickeners.
The general structural formula of monosaccharides, their building blocks, is CnH2nOn, but some monosaccharides do not fit this formula. Another name for carbohydrates is sugars. The name comes from the Greek word “gluco” meaning “sweet.”
Classification of Carbohydrates
Carbohydrates are divided into three groups according to their size;
1. Monosaccharides,
2. Disaccharides,
3. Polysaccharides
The smallest building blocks of carbohydrates are monosaccharides. Disaccharides are carbohydrates formed by the joining of two monosaccharides. Polysaccharides are carbohydrates formed by the combination of a higher number of monosaccharides.
The term oligosaccharide is composed of 2-10 monosaccharides in some sources. It is used for carbohydrates in some sources consisting of 2-20 monosaccharides.
1. Monosaccharides
Monosaccharides are the smallest building blocks of carbohydrates. When monosaccharides are broken down, a smaller carbohydrate is not formed. It consists of a single unbranched carbon chain. Chain lengths usually range between 2 and 8 carbons.
Oxygen is attached to one of the carbons in the chain with a double bond (C=O). Other carbons are bonded to the –OH group. If the carbon bonded to the oxygen by a double bond is at the end of the chain, such monosaccharides are “aldose”; if the double-bonded carbon is not located at the end of the chain, in other rows, such monosaccharides are called “ketose.”
For example, glucose is an aldose while fructose is a ketose. Therefore, while the closed formulas of the two compounds are the same (C6H12O6), their structures, physical and chemical properties are different from each other.
However, monosaccharides are also named according to the number of carbons in their chain length. To put it briefly;
- 3 carbon monosaccharides are “triose”; glyceraldehyde and dihydroxyacetone are trioses.
- 4 carbon monosaccharides are “tetrose”; erythrose and erythrulose are tetroses.
- 5 carbon monosaccharides are “pentose”; ribose, ribulose, arabinose and xylose are pentoses.
- 6 carbon monosaccharides are “hexose”; glucose, fructose, galactose, mannose and sorbose are each hexose.
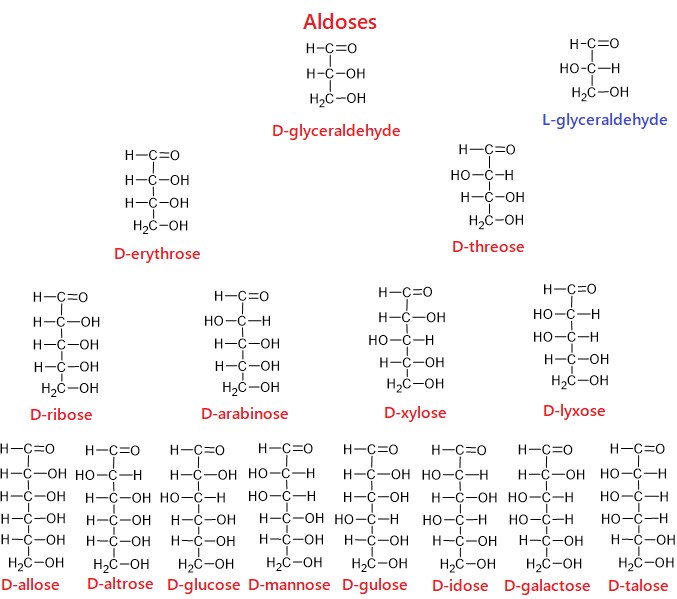
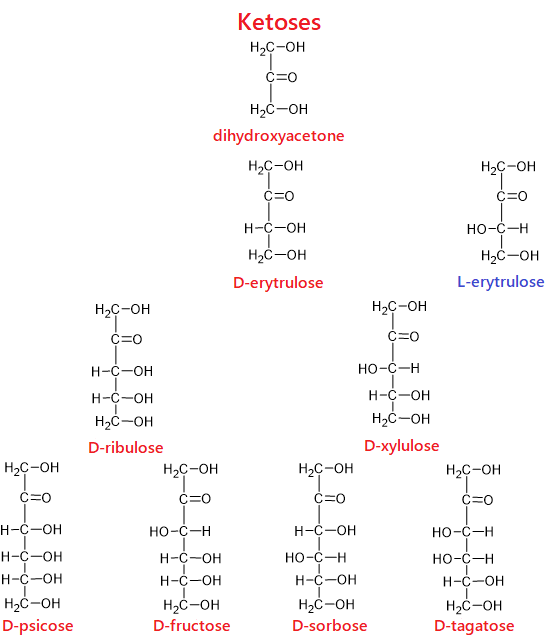
The molecular structure of monosaccharides and the topic of isomerism in monosaccharides are quite detailed subjects. Therefore, these topics are explained in detail in a separate article. In order to fully understand the subject of carbohydrates, I strongly recommend reading the article titled “Ring Structure, Conformations, and Isomerism in Monosaccharides.”
Common monosaccharides;
Ribose; is included in the structure of nucleotides, nucleic acids, B2 vitamins and enzymes. 2-deoxyribose, a derivative of ribose, is found in the structure of DNA.
Arabinose; is one of the building blocks of gums used as food additives. It is found in apples, figs, grapefruit and some grape varieties.
Glucose (Dextrose, blood sugar, grape sugar); is the most common monosaccharide in nature. It is found in free form in ripe fruits, honey and blood. Apart from that, they are primarily found in the structure of disaccharides and polysaccharides.
Fructose (Levulose, fruit sugar); is found in free form in sweet fruits and honey. It is the carbohydrate with the highest degree of sweetness among all carbohydrates. The absorption of fructose from the intestines is slower than glucose.
Galactose: It is not found in free form in nature; It is located in the structures of disaccharides and polysaccharides. The richest source is milk and dairy products containing lactose. Galactose is the building block of galactolipids found in the brain and nerve cells.
Mannose; It is not found free in nature. It is the building block of glycolipids, glycoproteins and serum albumins in animal tissues.
2. Disaccharides
The glycosidic bond of two monosaccharides forms disaccharides. They cannot be used directly on the body; They are broken down into their monomers in the digestive tract and the resulting monosaccharides are absorbed in the intestines.
Common disaccharides;
Sucrose; Sucrose consists of one glucose and one fructose molecule. Sucrose is found in all plants, but commercially available amounts are found in sugar beet and sugar cane.
Granulated sugar that we use as table sugar consists of sucrose crystals obtained from refining sugar cane and sugar beet.
Lactose (Milk sugar); It is found in nature only in the structure of milk. It is formed due to the combination of one molecule of glucose and one molecule of galactose.
Lactose is broken down into glucose and galactose by the enzyme lactase in the digestive tract. The absence or very little presence of lactase enzyme in the digestive system causes lactose to be indigestible. This situation causes a discomfort called “lactose intolerance” and manifests itself in bloating, pain, cramps, gas and diarrhea in the abdomen.
Maltose: It is a disaccharide that is not found in free form in nature and is generally obtained due to the hydrolysis of starch. It has two glucose molecules in its structure.
3. Polysaccharides
Structures formed due to the combination of many monosaccharides are called polysaccharides. There are polysaccharides consisting of 20 monosaccharides, as well as polysaccharides formed by the combination of 15,000 monosaccharides. Polysaccharides generally contain 200-3,000 monosaccharides.
Common polysaccharides;
Starch; Starch produced by plants to store energy is generally found in high structures such as seeds, fruits and roots. In addition to being an important energy source, starch is also used as a food additive in the food industry due to its properties, such as gelling and thickening.
Although there is only glucose as a building block in starch, glucose chains exist in 2 different forms: straight-chain (amylose) and branched-chain (amylopectin). The number of glucose in the starch structure varies according to which plant it is produced by; there are also plants in nature that produce starch molecules consisting of 3 million glucose.
Glycogen; They are polysaccharides consisting of glucose produced for energy storage in animal tissues and human tissue. It is not found in plant foods. While it is almost absent in the meat consumed, it can be found in the liver at a rate of about 6%.
Cellulose; Cellulose, which is found in the structure of cell walls in plants, consists of a very high amount of glucose. The importance of cellulose in human nutrition is that it is an excellent dietary fiber. This is because cellulose cannot be digested by humans. Although cellulose cannot be digested, it is a molecule that helps digestion as dietary fiber.
On the other hand, it is the raw material of paper and has a very high usage rate in the industry.
Here’s an article that might interest you; Carbohydrates; Functions in the Body and Daily Needs