Some Characteristics of Water
Water, one of the four fundamental components found in foods, is extremely important both because of its vital necessity in terms of nutrition and because of its influence on the physical, chemical, and microbiological properties of the food in which it is present.
In order to understand why water is so important, its main properties and characteristics must be known.
Water is a compound formed by the covalent bonding of two hydrogen atoms with an oxygen atom.
The shape of the water molecule and the fact that oxygen is more electronegative than hydrogen in O – H bonds give water a polar character with negative and positive ends. Some characteristics of water are provided below;
Hydrogen bonds are formed between water molecules, specifically between the oxygen of one water molecule and the hydrogen of a neighboring water molecule.
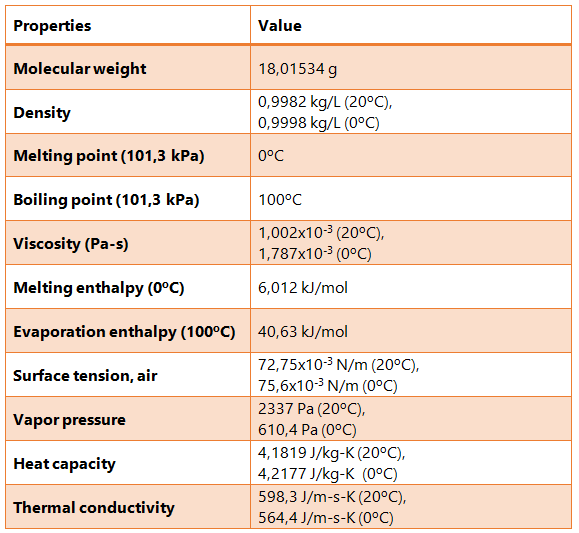
The fact that water molecules in a three-dimensional system form hydrogen bonds with one another results in water having high heat capacity, surface tension, melting point, enthalpy of vaporization and fusion, as well as strong adhesion and cohesion forces.
The high adhesion and cohesion forces of water enable it to easily adhere to and wet foreign substances and to move against gravity, as in the capillary vessels of plants.
The fact that the solid state of water (ice) has a lower density than its liquid state is also related to the number of intermolecular hydrogen bonds formed and the lengths of these hydrogen bonds.
The presence of positive and negative poles in the water molecule allows ionic compounds to dissolve well in water. In addition, non-ionic but polar compounds such as sugars and simple alcohols can also dissolve in water.
The effect here mainly arises from the formation of hydrogen bonds between polar groups (between the oxygen of water and the carbonyl group of the dissolved compound).
Forms of Water in Foods
All foods contain water in varying amounts. For example, meat consists of about 70% water, and milk consists of approximately 87% water. The table below shows the water content of some foods;

However, not all the water present in foods behaves like water or possesses all of water’s properties. Water exists in foods in five different forms. These forms are as follows;
1. Structural Water: Water that exists as part of components other than water. It has no solvent character, does not freeze at –40°C, and its water activity is zero. It constitutes a very small percentage of the total water in foods.
2. Vicinal Water: Water bound to compounds with strong hydrophilic polar groups. It has no solvent character, does not freeze at –40°C, and its water activity is zero. In high amounts, it can surround hydrophilic polar groups in a single layer.
3. Multilayer Water: Water bound to vicinal water. Due to its proximity to non-water components, it may partially exhibit the characteristics of pure water. A large portion does not freeze at –40°C and may exhibit solvent properties.
4. Free Water: Water in food that is distant from non-water components and interacts only with other water molecules. The characteristics of this water resemble those of dilute salt solutions. Free water can freeze below 0°C and can exhibit solvent properties.
5. Entrapped Water: Water located between large macromolecules. It can be easily removed by drying or easily frozen. Water in pectin and starch gels, as well as in animal and plant tissues, can be given as examples of entrapped water.
Based on this classification, it can be said that not all the water present in a food exhibits the properties of water. This means that not all the water contained in a food is “available” for physical and chemical reactions and for the activity of microorganisms.
Therefore, in addition to the total amount of water in a food, knowing how much of that water exhibits the properties of water is extremely important for food preservation and processing. One of the reasons why two different foods containing the same proportion of water may have different shelf lives is the difference in the proportions of the various water forms they contain.
The parameter known as “water activity” is used to measure the “activity” of the water in the foods. Water activity is defined as the ratio of the vapor pressure of water in a food to the vapor pressure of pure water at the same temperature.
The symbol of water activity is “aw.” Water activity ranges between 0 and 1 and is expressed without units. For example, the water activity of pure water is 1, that of honey is approximately 0.60, and that of milk powder is approximately 0.2.
The importance of water activity can be explained as follows;
Most harmful bacteria require an environment with at least 0.91 aw to sustain their life activities. If the water activity of a food is below 0.91, or if the food is processed (such as by drying or concentrating) to reduce its water activity below this value, the food is no longer suitable for the growth of most harmful bacteria. Therefore, these bacteria can no longer cause harm to the food (this topic will be examined in more detail in the food microbiology section).
From the perspective of chemical reactions, the Maillard reaction reaches its maximum rate at aw values between 0.65 and 0.70. In foods containing high amounts of amino acids and reducing sugars, the water activity value is highly important. Similarly, the amount of vitamin C present in a food is closely related to its water activity value. As water activity increases, the rate of vitamin C degradation also increases.
However, it should also be noted that while the water activity parameter is effectively used at temperatures above the freezing point, it largely loses its validity at temperatures below the freezing point. (For more detailed information see Water Activity in Foods; Definition and Effects on Microbial Activities).
Another important concept in the relationship between foods and water is “equilibrium moisture.” As is known, foods absorb moisture from or release moisture to their surrounding environment.
While a biscuit in the same environment absorbs moisture and becomes soft, a tomato loses moisture and dries. After a certain period of time, the moisture content of the food and that of the surrounding environment reach equilibrium and stabilize.
The point at which equilibrium is reached is called the “equilibrium moisture content” and is expressed in units of g moisture/g dry matter. Naturally, the equilibrium moisture content varies depending on the temperature and relative humidity of the environment.
When the process of a food reaching its equilibrium moisture content as a result of moisture exchange with the environment is represented graphically, “sorption isotherms” are obtained. The graphical representation of a completely dry food gaining moisture from the environment until equilibrium is reached is called the “adsorption isotherm.”
The graphical representation of a moist food losing moisture until it reaches equilibrium with the environmental humidity is referred to as the “desorption isotherm.” Data for sorption isotherms are obtained experimentally. Equilibrium moisture content and sorption isotherms are extremely important in the storage and preservation of foods.
Here are some articles that might interest you;
Water and Health; Functions in the Body and Daily Needs
Quality Characteristics That Drinking Water Should Have